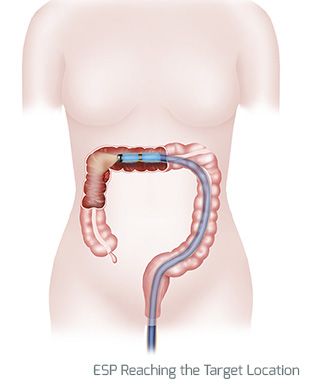
MINT has developed the Endolumenal Surgical Platform (ESP), a revolutionary medical device that will enable GI surgeries to be performed endolumenally – entirely from within the channel of the intestine – reducing anesthesia, shortening hospital stays, reducing healthcare costs, accelerating patient recovery, and improving clinical outcomes. ESP is the first step in MINT’s goal to migrate all GI surgeries to endolumenal procedures, opening an entirely new field of endolumenal GI surgery.
Unmet Clinical Need
- Endolumenal Intestinal Surgery: Less invasive intestinal procedures are needed to treat the rapidly expanding GI patient population. Patients who cannot withstand traumatic surgical interventions or general anesthesia would greatly benefit from endolumenal alternatives to open or laparoscopic surgeries.
- Surgical Platform and Tools: Clinicians require a platform that leverages current endoscope capabilities (including HD imaging, irrigation, suction, inflation, etc.) but provides better surgical control and precision to enable endolumenal procedures to be performed using a set of dedicated tools.
- Stability: Currently, clinicians cannot fix endoscopes into position relative to the intestinal wall, making the endoscope tip difficult to control and endoscope-based precise surgery almost impossible due to colon peristalsis and slips.
- Visualization: Advanced diagnosis and endolumenal procedures are currently limited by the lack of visibility in the colon, especially around flexures and behind folds. As a result, up to 41% of polyps are missed during colonoscopy because of poor visibility. A device that can improve the visualization provided by current endoscopes would enable better diagnosis and advanced endolumenal therapeutic procedures to be performed for the first time in difficult locations.
Solution
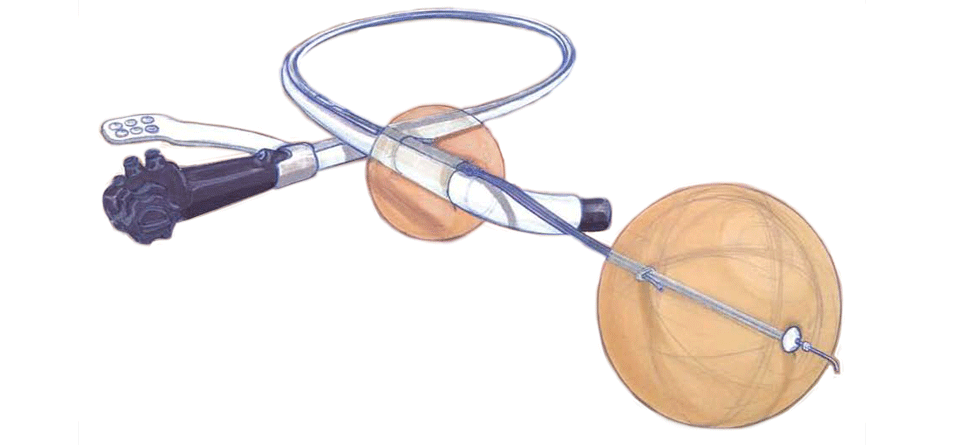
ESP is a stabilized endolumenal platform that expands the capabilities of currently available tools and enables complex minimally invasive endolumenal procedures.
- ESP is a disposable device that fits over commercially available endoscopes, dramatically improving diagnostic and therapeutic capabilities inside the channel of the intestine without inhibiting any current endoscope functionality.
- ESP’s dual balloon system creates a stabilized, isolated diagnostic and therapeutic- zone within the intestine, allowing clinicians to visualize, manipulate, and treat isolated segments of bowel.
- ESP’s forward assembly allows clinicians to precisely extend and retract ESP’s forward balloon and manipulate the colon between the two balloons that define the therapeutic zone, reducing the angle of bends and flattening folds in the colon to expose lesions for diagnosis and treatment and improving visualization.
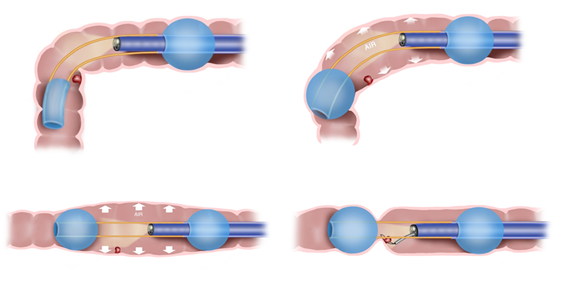
Figure: ESP passing the flexure with the aft balloon (top left), Improved visualization in the flexure (top right), Established Therapeutic Zone (lower left), Manipulating bowel to optimize polyp access for treatment (lower right)
Study Results

Proof of Concept In Vitro: Improved Visualization - (40% increase in visible area)
In the hands of colorectal surgeons and interventional endoscopists, ESP as designed, working in conjunction with currently available endoscopes and instruments, will enable advanced procedures to be performed endolumenally. This will dramatically improve outcomes compared to open or laparoscopic procedures by reducing surgical trauma and accelerating recovery, meanwhile driving significant cost out of the healthcare system. ESP-enabled procedures will spare 200,000 patients per year from invasive surgical procedures to treat their difficult polyps, and ESP could save $650M to the US Healthcare system per year just for the treatment of difficult polyps.
ESP in its current form is MINT’s first step in enabling complex endolumenal surgeries for a wider range of target applications. Pipeline developments include advanced tools that will leverage ESP’s stable platform and provide better surgical capabilities. This will enable the development of endolumenal approaches for additional therapeutic areas such as diverticular disease, stricture, intestinal fistulae, prolapse, volvulus, anastomosis leaks and early cancers.